
What is alectinib (Alecensa) and why is the latest news a positive step for lung cancer patients?
Alectinib, which is known under the brand name Alecensa, targets a specific type of lung cancer: metastatic non-small cell lung cancer (NSCLC) with anaplastic lymphoma kinase positive (or ALK-positive) gene, and may have benefits for patients affected by this type of cancer. It was approved by the Japanese medical agency (PMDA) in 2014 and shortly after by the American FDA, the European EMA and the Australian TGA.
Alectinib (Alecensa) is indicated for patients who are anaplastic lymphoma kinase positive (ALK-positive), which means they have a mutation in the ALK gene. This gene is thought to play an important role in how NSCLC grows and spreads to other parts of the body, which is why new treatments have been developed that specifically target this gene. These medicines are called ALK-inhibitors and alectinib (Alecensa) is one such medicine. Although it is not globally available, patients to whom it is not accessible can import this medicine into their country, safely and legally.
Alectinib (Alecensa) is an ALK-inhibitor used to treat metastatic non-small cell lung cancer (NSCLC) in patients who have a particular gene mutation. It was initially approved as a second-line treatment for patients whose cancer progressed on or after a first treatment. Its approval has recently been extended to include the treatment of patients whose metastatic cancer has not been treated yet (first-line treatment). It is an oral medicine, taken in capsules.
Learn more about costs and delivery
Alectinib (Alecensa) may be beneficial for patients who have ALK-positive NSCLC and have not been treated for their metastatic disease yet. A study, which involved 303 patients, compared alectinib (Alecensa) with crizotinib (another drug for ALK-positive NSCLC).
People taking alectinib (Alecensa) lived twice as long without their disease worsening when compared to crizotinib. Of the patients with brain metastases, those taking alectinib (Alecensa) responded better than those who were taking crizotinib. The brain metastases completely disappeared in many of the patients who were taking alectinib (Alecensa).
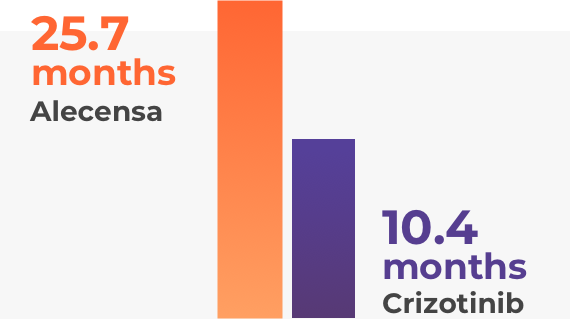
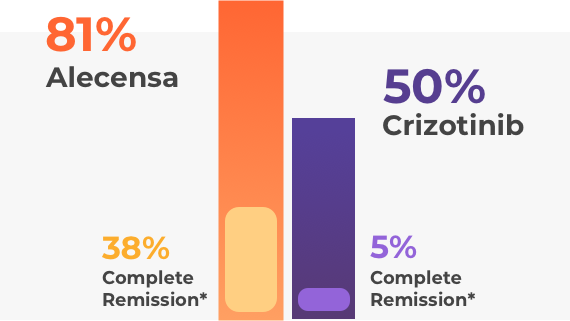
Previous studies have proven the efficacy of alectinib (Alecensa) in patients with metastatic ALK-positive NSCLC who had previously received treatment for their metastatic diseases.
*Complete response (CR or complete remission) means that the cancer has responded to the treatment and no evidence of cancer can be detected. Partial response (PR or partial remission) implies that although an effect of the treatment is evident, further treatment will probably be required to attempt a complete response.
For more details refer to the prescribing information and references at the bottom of the page.
The most common side effects include tiredness, constipation, oedema (accumulation of fluids), muscle pain and anemia (lack of red blood cells). Serious adverse reactions include drug-induced liver injury, interstitial lung disease (ILD)/pneumonitis, kidney failure, slow heart rate, severe muscle pain, and muscle tissue injury.
Please note this is not intended to be a comprehensive guide. Consult your treating doctor and the prescribing information at the bottom of the page for full details of side effects.


Please note this is not intended to be a treatment plan. For a personalised treatment plan consult your doctor. For more details, you can also reference the full prescribing information at the bottom of the page



For more information about the way alectinib (Alecensa) works, consult your doctor or see the prescribing information at the bottom of the page. This is not intended to be a comprehensive scientific explanation.

If you or one of your relatives or friends are a patient with ALK-positive NSCLC, you might want to ask questions about the medicine for this disease. If you’d like to ask questions about how to get alectinib (Alecensa), or about our service in general, our team of qualified pharmacists and experts are ready to offer support and assist with queries. Our team speaks 17 different languages and is ready to help.
everyone.org makes the latest approved medicines available to patients, hospitals and doctors around the world, in a legally compliant way. We source innovative medicines at the best possible prices and ensure safe and efficient delivery. We have successfully delivered to over 88 countries worldwide, and have already supported over 11,000 doctors and patients.
Find out more